|
1/18/2024 0 Comments Polypeptide backboneAnd here, you have a nitrogen atom, the alpha carbon, and a carbonyl carbon. Now, if we take a closer look at the backbone of this chain, we can see that there is a pattern formed by the atoms that form this backbone. 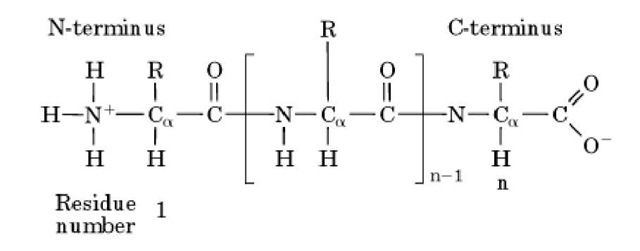
And if we kept adding amino acids along in a chain here, we would have a polypeptide. The terms phi and psi refer to rotational angles about the bonds between the N-alpha carbon and alpha carbon-carbonyl carbon respectively (previous page). So now, here we can see we have a dipeptide. Peptide bond is planar: twisting can occur around the alpha carbon, but not around the peptide bond. Another consequence of considering the peptide bond as a double bond is that it reduces the number of variable rotational angles of the polypeptide backbone. These hydrogen bonds occur at regular intervals of one hydrogen bond every fourth amino acid and cause the polypeptide backbone to form a helix. Cells use only 20 amino acids to make polypeptides and proteins, although they do use a few additional amino acids for other purposes. Peptide bond is polarised and polar groups can H bond. helix: In an -helix, the polypeptide backbone forms a repeating helical structure that is stabilized by hydrogen bonds between a carbonyl oxygen and an amine hydrogen. The primary structure of a protein refers to the amino acid sequence of its polypeptide chain (s). In our model, referred to as a Peppytide, we emulate this basic structure of the polypeptide chain backbone by linking two types of units together: the amide units and the -carbon units (C ), connected alternately at. Helical wheel projections can be made showing the polarity of the faces of the helix looking down the axis.\)). L amino acids and the C-N-C-N- polypeptide backbone. Thus, the polypeptide backbone can be represented as an alternating copolymer of the amide unit and the -carbon unit. Each type of protein differs in its sequence and. 
can form when the 2 or 3 alpha helices have most of their nonpolar side. A peptide bond forms when the carboxylic acid group (R-COOH) of one amino acid reacts with the amine group (R-NH 2) of another.The resulting molecule is an amide with a CN bond (R-C(O)-NH-R). common structural motif in proteins in which different sections of the polypeptide chain run alongside each other and are joined together by hydrogen bonding between atoms of the polypeptide backbone. R-groups may also be fully hydrophobic within alpha helices that span the plasma membrane as shown in (D). A protein consists of a polypeptide backbone with attached side chains. the repeating sequence of atoms along the core of the polypeptide chain. When multiple strands from different regions of a polypeptide interact in this way Figure 2 The crystal structure of a synthetic glycine polymer suggested that folded proteins feature hydrogen bonds between backbone. As few as two strands can interact in this way. In amphipathic helices, hydrophilic residues are positioned on one side of the helix and hydrophobic on the other as shown in the side view (A) or top-down views (B & C). Instead, hydrogen bonds form between the backbone atoms of separate -strands. Figure modified from: Khara, J.S., et al.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
